
Home » Jubilant HollisterStier broadens its expansion plans in East Spokane
Jubilant HollisterStier broadens its expansion plans in East Spokane
Pharmaceutical company to double capacity by 2024
June 17, 2021
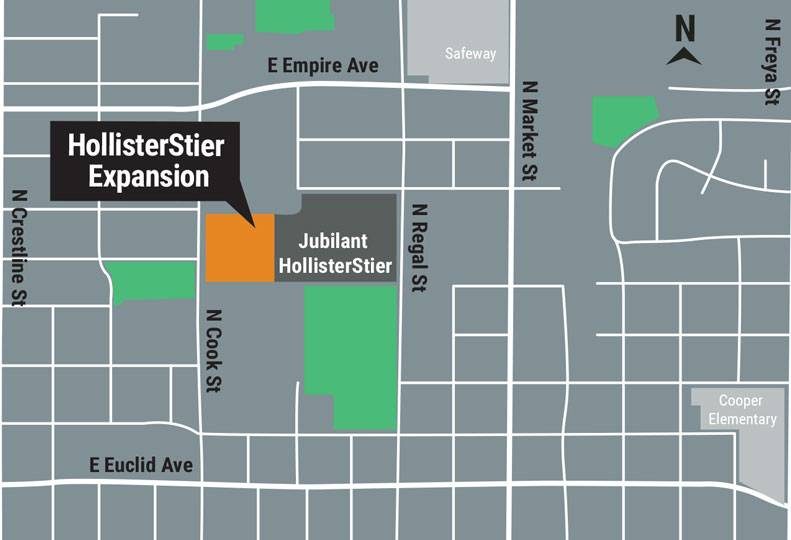
Jubilant HollisterStier plans to invest $92 million to expand its existing facilities in North Spokane and double its capacity, broadening the scope of an expansion the company first disclosed late last year.
With the project, the company plans to add over 50,000 square feet of space to its facility at 3525 N. Regal.
The company announced the project updates in a press release, and a representative of the company didn’t immediately expand on company plans.
Plans include adding a new high-speed 400-vial-per-minute injectable fill line, with isolator technology and two 300-square-foot lyophilizers, or freeze dryers, which remove water to extend shelf life or make material more convenient for transport.
The company currently has two 385-square-foot lyophilizers and two 220-square-foot lyophilizers.
The isolator technology allows pharmaceutical companies to reach high levels of sterility.
Amit Arora, Jubliant HollisterStier president, says in the release, “We are excited to expand our sterile injectable and vaccine manufacturing capacity and meet the increasing demand from our customers.”
Arora told the Journal in December that the expansion, then estimated at $70 million and 120,000 square feet, would add 400 employees over the next three years.
As of December, Jubilant HollisterStier had 705 employees.
Integrated Project Services LLC, a Pennsylvania-based pharmaceutical manufacturing consulting company, designed the expansion.
Jubliant HollisterStier has begun hiring for the new positions, according to the release.
The project is expected to be completed by the end of 2024.
Jubilant HollisterStier is the contract manufacturing arm of Yardley, Pennsylvania-based Jubilant Pharma Ltd. The company has about 5,200 employees worldwide across six U.S. Federal Drug Administration-approved manufacturing facilities in the U.S., Canada, and India, as well as a network of 49 radiopharmacies, which are pharmacies that specialize in preparing radioactive materials for specific diseases, in the U.S.
“With two facilities in North America working to manufacture multiple COVID-19 vaccines and therapies, we remain committed to supporting efforts to eradicate this global pandemic and are prepared for a potential future pandemic response,” says Jubilant Pharma CEO Pramod Yadav. “This expansion will also help us meet high demand from our global specialty pharmaceutical companies, with whom the company has established strong relationships.”
Jubilant HollisterStier also announced in March that the company is working with Novavax, an American biotechnology company, to provide fill-finishing manufacturing services for the production of the company’s COVID-19 vaccine candidate. Novavax announced this week that the vaccine demonstrated 90.4% overall efficiency and 100% protection against moderate and severe disease in its clinical trial across the U.S. and Mexico.
On June 15, Ocugen Inc., a biopharmaceutical company that develops gene therapies to cure blindness diseases and is working to develop Covaxin, a COVID-19 vaccine candidate, announced it has partnered with Jubilant HollisterStier to manufacture the vaccine for the U.S. and Canada.
Covaxin is India’s COVID-19 vaccine that was developed by Bharat Biotech in collaboration with the Indian Council of Medical Research-National Institute of Virology.
Like this story?
You’ll love the rest. Subscribe today, and you’ll receive a year’s subscription to the Journal of Business, unlimited access to this website, daily business news emails, and weekly industry-specific
e-newsletters. Click here for 50% off your first year.
Latest News Health Care Manufacturing Technology




